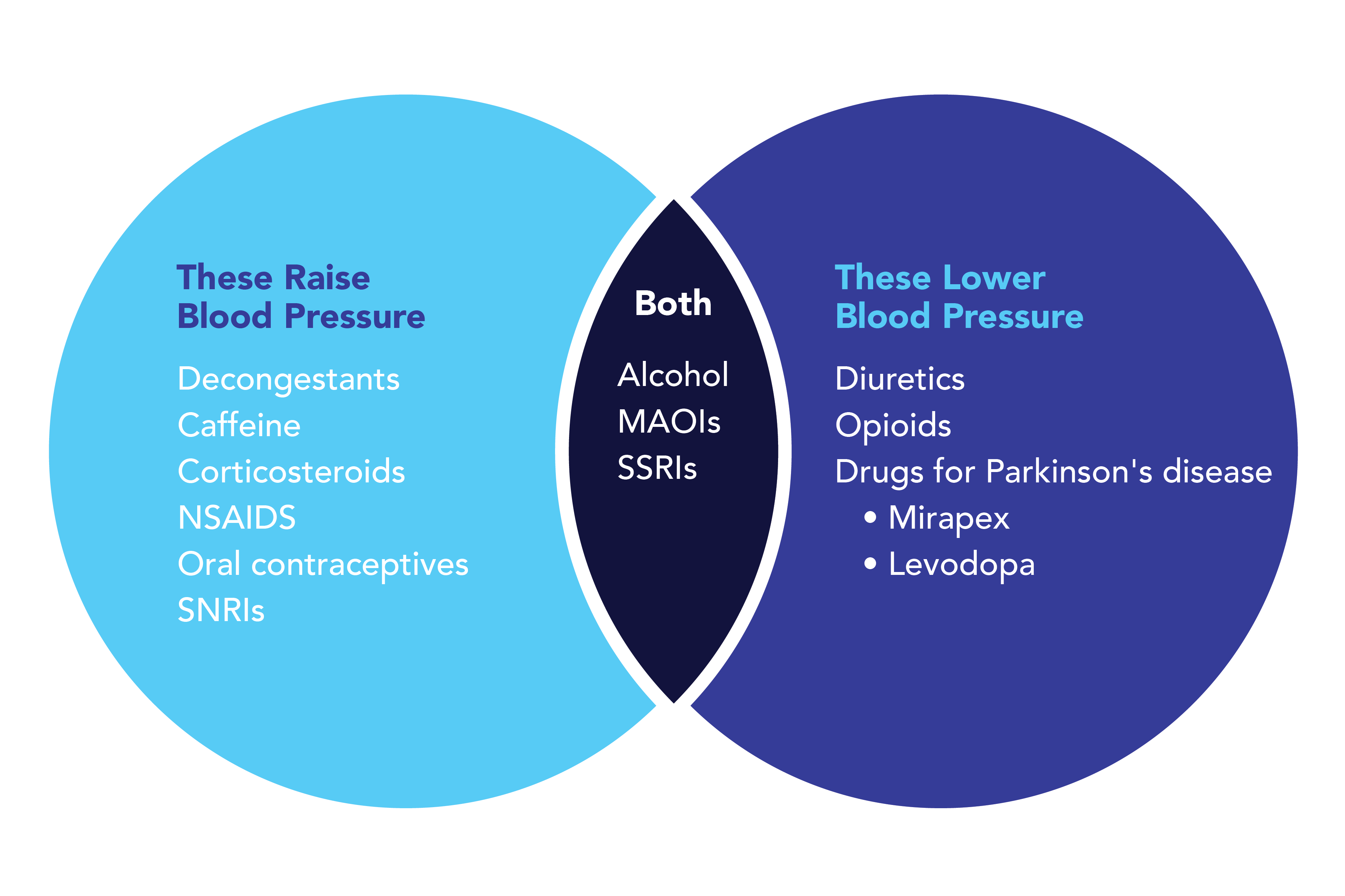
Early in 2022, the FDA granted emergency use authorization to two antiviral drugs to treat COVID-19: Lagevrio (molnupiravir) and Paxlovid (nirmatrelvir and ritonavir). These treatments aren’t 100% effective, though they are more effective the sooner they’re prescribed, so it’s been crucial for scientists to continue to study other drugs and combinations to determine the best treatments—old and new—for COVID-19.
Since the start of the COVID-19 pandemic, researchers and the biopharmaceutical industry have been working to determine if available medications can treat COVID-19, knowing that existing treatments would be faster and easier to get to people who need them than newly-designed drugs.
Some of the treatments they’ve tried have failed, while others have proven useful in certain scenarios, like if you’re hospitalized. Researchers have put together the data, and one group has created a registry of more than 350 drugs being tested to treat the disease or its symptoms. Additionally, on March 15, 2021, the Food and Drug Administration (FDA) launched the FAERS (FDA Adverse Event Reporting System) public dashboard. There, patients, healthcare providers and manufacturers can report and review adverse events associated with drugs and therapies that have received emergency use authorizations (EUAs) for COVID-19. Vaccines are not included; adverse reactions to vaccines are reported through the Vaccine Adverse Event Reporting System (VAERS.)
Here are the highlights of what researchers have found:.
A Two-Pronged Approach
One approach to treat a COVID-19-related illness is to find medications that slow down — or even stop — the virus’s ability to reproduce. This could shorten the illness’ duration and prevent it from becoming severe or critical.
Researchers are also investigating drugs that reduce the body’s immune response in the critical stages of illness. An overzealous immune response can cause inflammation that damages cells and organs throughout the body.
There isn’t enough scientific evidence about the safety or effectiveness of most of these drugs for them to have garnered FDA approval to treat COVID-19. Larry Sasich, PharmD, MPH, a consultant for the FDA and the Saudi Arabian Food and Drug Authority, and co-author of Worst Pills, Best Pills, also pointed out that, for the most part, we only really know about side effects in patients who don’t have COVID-19.
Be sure to talk with your doctor about any medication that you’re considering using to treat COVID-19. Not only is there the possibility for dangerous side effects, but there are also potential drug interactions to consider that require medical supervision.
Potentially Promising Treatments
Here’s a look at some of the drugs that scientists are researching to see if they could be effective in treating COVID-19-related illnesses.
Antacid
What it’s currently used for: Pepcid (famotidine) is currently used as a daily pill to prevent acid reflux. It reduces your production of stomach acid, and researchers believe, of certain inflammatory proteins called interferons.
Clinical Trials: Some early studies suggested the drugs helped patients who were hospitalized but another large study contradicted those findings. Researchers published the only randomized clinical trial in the journal Gut, in February 2022, which showed that high doses of the drug (80mg, while a typical pill contains 20mg) taken three times a day for up to 14 days reduced the duration of symptoms in non-hospitalized patients. The study only included 55 patients and larger studies are required to validate these results.
Known serious side effects: Famotidine is generally considered safe when used short-term, though longer term use and higher doses do come with a variety of serious side effects such as an elevated risk for kidney problems and cognitive impairment.
Antidepressants
Fluvoxamine
What it’s currently used for: Fluvoxamine is a selective serotonin reuptake inhibitors (SSRI) commonly used to treat obsessive-compulsive disorder (OCD.) SSRIs are also used to treat depression. It increases the availability of a the neurotransmitter serotonin. Fluvoxamine also reduces inflammation, which is how it may help with COVID-19.
Clinical trials: There were several small trials early in the pandemic that suggested the drug might help reduce hospitalization and death, but on October 27, 2021, a much larger study of 1500 patients was published in The Lancet. The trial showed that the drug reduced hospitalizations by two-thirds, when taken as directed. There was only one death in the fluvoxamine group, compared to 12 in the placebo group.
Known serious side effects: The phrase “taken as directed” was crucial here, as 84 patients in the trial discontinued using the drug due to intolerability. (However, 64 also stopped using the placebo as well.) When these patients were accounted for, fluvoxamine only decreased hospitalizations by one-third. The most common side effects are constipation, headache, difficulty sleeping and fatigue. However, there are many concerning less common side effect such as feeling of constant movement of self or surroundings, abdominal pain, twitching, trouble breathing, trouble urinating, blurred vision and bruising. Researchers told the New York Times that the intolerability suggested that the researchers may not have found the ideal dose yet.
IL-6 Receptor Inhibitors
The IL-6 (interleukin-6) receptor inhibitors tocilizumab and sarilumab are both injections and “they are quite potent in reducing inflammation,” said Hana Akselrod, MD, an assistant professor of medicine in infectious diseases at the George Washington University School of Medicine and Health Sciences in March 2020.
How IL-6 inhibitors could help COVID-19 patients: The theory is that these medications “try to block parts of that severe immunologic inflammatory cascade in order to reduce the severity of the pneumonia that happens after the virus (COVID-19) has already caused the infection,” said Akselrod.
“There do appear to be some data that suggest that [Il-6 inhibitors] might be effective as adjuncts to treatment in patients with severe COVID-19 pneumonia, reducing some of the inflammation and some of the other complicating factors, hopefully, to improve oxygenation,” said Michael Klepser, PharmD, FCCP, a professor at Ferris State University’s College of Pharmacy, in March. “They’re not going to necessarily cure the virus, but they may help patients improve clinically.”
Tocilizumab (Actemra/RoActemra)
What it’s currently used for: Tocilizumab is FDA-approved to treat active rheumatoid arthritis (RA), giant cell arteritis, juvenile idiopathic arthritis and cytokine release syndrome. It works by decreasing the overactive inflammatory response found in these conditions.
Clinical trials: Researchers have conducted several trials yielding mixed results. The National Institutes of Health (NIH) recommended against their use this summer. However, a new study posted on a preprint server suggested that both tocilizumab and sarilumab may reduce the death rate of patients in the intensive care unit from 36% to 27%. Scientists note that the main difference between this study and previous ones is that the current study was conducted only in critically ill patients.
The NIH now recommends that the drug be used in combination with dexamethasone to treat certain hospitalized patients with severe COVID-19.
Known serious side effects: Tocilizumab has multiple potential side effects, the most consequential is an increased risk of developing serious infections that can lead to hospitalization or death. Because of that risk, tocilizumab has a black box warning. “A boxed warning is the most serious warning that the FDA can require in the professional product label for a drug,” said Sasich. “Usually, it involves deaths in human beings.” Other severe adverse reactions include tears in the stomach or intestines, liver issues, low platelet count, higher blood-cholesterol levels, low neutrophil (white blood cell) count, allergic reactions and an increased risk of cancer.
Sarilumab (Kevzara)
What it’s currently used for: Sarilumab (Kevzara) is FDA-approved to treat active RA, particularly in people who haven’t responded to other medications for RA or who can’t use other types of medication.
Clinical trials: Early on, researchers conducted several trials yielding mixed results. However, a study posted on a preprint server in January 2021 (meaning it was not yet peer-reviewed, a process used by the best publishers to ensure high-quality research was conducted) suggested that the both tocilizumab and sarilumab may reduce the mortality rate of patients in the intensive care unit from 36% to 27%. Scientists note that the main difference between this study and previous ones is that the current study was conducted only in critically ill patients.
The NIH suggests using sarilumab in combination with dexamethasone for severely ill, hospitalized COVID-19 patients when tocilizumab is unavailable.
Known serious side effects: Sarilumab, too, has a black box warning about an increased risk of developing serious infections that can lead to hospitalization or death. It also has the same potential grave adverse reactions as tocilizumab, including tears in the stomach or intestines, liver issues, low platelet count, higher blood-cholesterol levels, low neutrophil (white blood cell) count, allergic reactions and an increased risk of cancer.
Antiviral Drugs
Remdesivir
Remdesivir was originally formulated to fight the Ebola virus. However, it was found to be basically ineffective against Ebola during the 2019 outbreak in the Democratic Republic of the Congo.
What it’s currently used for: Remdesivir works by inhibiting a virus’ ability to replicate, but it wasn’t approved to treat any type of condition or disease prior to the pandemic.
Clinical trials: Remdesivir was the first (and only) drug fully approved by the FDA to treat COVID-19 in October 2020, though it had received an EUA before that in May. It was one of the drugs used to treat former President Donald Trump when he contracted the illness. Trials showed that the drug shortened recovery times from 15 to 10 days for certain hospitalized patients and that the drug was most effective in those who were receiving oxygen but were not ventilated. The NIH now recommends that the drug be used in these patients.
Known serious side effects: Not a lot of information is available for this drug because it wasn’t previously approved. “One concerning bit about it is it does have a solubilizing [dissolving] agent in it called SBECD (sulfobutylether-beta-cyclodextrin),” said Klepser. “We’ve seen that solubilizer used with other medications and sometimes in patients with renal dysfunction [it] can cause some problems because (the SBECD) can accumulate and cause kidney damage. That would be the major thing to keep an eye on.”
The NIH lists allergic reactions, nausea and liver damage as possible side effects, but it’s noted that there may be more we don’t yet know about.
How it could help COVID-19 patients: In 2017, researchers found that remdesivir was effective against many different coronaviruses — including the ones that cause severe acute respiratory syndrome (SARS) — in both mice and in cell cultures.
“The idea is treating with remdesivir would prevent the viral infection from becoming established and would also shorten its duration once it’s already happening,” said Akselrod.
Ritonavir and Lopinavir (Kaletra)
Ritonavir and lopinavir are antivirals used in a combination drug called Kaletra.
What it’s currently used for: Kaletra is FDA-approved to treat human immunodeficiency virus (HIV). This medication blocks the virus’s ability to copy its genetic material, said Akselrod. “This is a medication we’ve been using for many, many, many years to treat HIV, and it’s a potent and effective antiviral, but for a virus that’s not in the same family as COVID-19,” she said.
Clinical trials: Trials have not demonstrated that the drug helps patients with COVID-19. The NIH recommends against their use for COVID-19.
Known serious side effects: There are a number of other medications that shouldn’t be taken with Kaletra because of the risk of acute drug interactions. Kaletra use can also lead to pancreatitis, liver damage, heart rhythm abnormalities, increased cholesterol levels and new or worsened diabetes.
Anti-Parasitic Drugs
Chloroquine and Hydroxychloroquine
What they’re currently used for: Both chloroquine and hydroxychloroquine medications are FDA-approved to treat and prevent malaria. “Remember that malaria is not a virus or a bacterium, it’s a parasite,” said Sasich. Because parasites have cells that are similar to human cells, a drug that’s used to treat a parasitic infection like malaria may cause more serious adverse effects, he says.
Chloroquine is also approved to treat amoebiasis, a gastrointestinal infection caused by an amoeba. Hydroxychloroquine is approved for systemic lupus erythematosus and RA in patients whose symptoms haven’t improved using other medications.
Clinical trials: On March 30, 2020, the FDA granted chloroquine and hydroxychloroquine an EUA for the treatment of COVID-19. While clinical trials were still ongoing, the approval was made so that the government could add it to the Strategic National Stockpile and it would be available for hospitals to request and to supply for clinical trials. However, in June, after the trials ended and demonstrated that the drug did not benefit patients with COVID-19, the authorization was revoked.
In a November 2020 study in the New England Journal of Medicine, scientists reported that the drug also does not prevent COVID-19 when taken after close contact with an infected person.
Known serious side effects: Chloroquine’s potential adverse effects include cardiomyopathy (heart muscle disease) that can result in heart failure, heart rhythm abnormalities, severe hypoglycemia (low blood sugar), permanent eye damage, muscle weakness, gastrointestinal distress, neuropsychiatric disorders and immune system dysregulation. Using chloroquine with certain other medications can lead to serious drug interactions.
Hydroxychloroquine has a risk of similar serious side effects, such as permanent eye damage, cardiac effects like cardiomyopathy with a risk for heart failure and heart rhythm abnormalities, severe hypoglycemia and muscle atrophy. It can also cause suicidal behavior. Other grave effects have been reported as well, including disorders of the blood, heart, ear, eye, immune system, metabolism and skin.
How antimalarials were thought to help COVID-19 patients: Akselrod said the theory was “essentially (that) those drugs would make the cell less hospitable to the virus by interfering with some of the mechanisms that are involved in both the virus entering the cell and then possibly with the inflammation that occurs around the infection.”
Like remdesivir, chloroquine and hydroxychloroquine have been found to be effective against coronaviruses in cell cultures, including ones that cause SARS, but not in humans with COVID-19.
Potential risks for COVID-19 patients: Not everyone can take these drugs, Akselrod said. “Chloroquine and hydroxychloroquine can cause quite severe poisoning and even fatalities, if they’re not taken under the supervision of a doctor or if they’re ingested in the wrong dose or by someone with a heart condition that’s predisposed to an irregular rhythm. These drugs can trigger an unstable heart rhythm, which can cause the heart to stop, which is why it’s so concerning that right now there seems to be a worldwide rush on these drugs.”
“The major concerns with these medications are their toxicities and that when they’re used, some patients experience very severe side effects, including cardiovascular conduction abnormalities and agranulocytosis, which is kind of like wiping out your immune system,” Klepser said.
People who hoarded these drugs or took them unsupervised became a problem, too, said Akselrod. “This is in the context of an unprecedented global emergency, and it’s quite understandable if people are scared and trying to reach for anything that’s available that we think might help. But people who depend on these drugs to control their lupus or [RA] find themselves unable to refill their prescriptions. They’re at risk of having their disease flare up and force them to go to the hospital at a time when it’s very unsafe for them to be doing that and when they should be isolating at home and trying to avoid infection at all costs.”
Ivermectin
What it’s currently used for: Ivermectin is used to treat parasitic worms. It comes as pills or as topical creams to treat head lice and other skin conditions. One formulation is commonly used in dogs to prevent heartworm. The FDA has warned consumers not to self-treat with ivermectin, especially with formulations meant for animals.
Clinical trials: In June 2020, researchers published a study showing that the drug could prevent the virus that causes COVID-19 from replicating in Petri dishes. If it worked the same way in humans, this would be likely to significantly reduce symptom severity. Some doctors started prescribing it to COVID-19 patients, especially in Latin America, but the NIH warned that, to be effective, it may be required to use incredibly high doses (up to 100 times the usual anti-parasitic dose), which would create new safety concerns. The NIH for now has concluded that there was not enough evidence to recommend for or against its use. Several clinical trials have tested the drug in COVID-19 patients, but results have been inconsistent.
One popular study that supported the drug’s use was later withdrawn due to poor methodology and plagiarism. A separate trial, the Together clinical trial, stopped testing ivermectin after interim analyses suggested the drug provided no benefit.
A Cochrane Review published on July 28, 2021, determined that there was not enough evidence to demonstrate that ivermectin is effective against COVID-19, and that almost all completed studies were of low quality.
Some clinical trials are still ongoing, and organizations, like the World Health Organization (WHO) and the Infectious Disease Society of America, say that only patients enrolled in these trials should be taking ivermectin for COVID-19. The drug’s manufacturer Merck does not recommend it be used to treat the disease.
Known serious side effects: Calls to poison control and emergency room visits by patients who have ingested ivermectin skyrocketed over the summer of 2021, according to a bulletin from the American Association of Poison Control Centers. The drug can cause headaches, dizziness, muscle pain, rapid heart rate, chest discomfort, shaking, nausea and diarrhea. Rarely, patients can also have severe allergic reactions.
Many people have tried to use formulations designed for horses and cows. These are far more potent than the drugs prescribed to humans and can cause extremely dangerous side effects like hallucinations and seizures, according to the Centers for Disease Control and Prevention (CDC).
Interactions with other drugs: Blood thinners and ivermectin strengthen each other’s impacts, increasing the likelihood of dangerous side effects. The drug can also interact with supplements like St. John’s wort, experts told NPR.
Convalescent Plasma
What it’s currently used for: In August 2021, the FDA announced an EUA for convalescent plasma. It’s thought that since the blood of patients who have recovered from COVID-19 should be filled with antibodies, giving a portion of it to current patients should help them recover. It’s the same idea behind the monoclonal antibodies, which are mixtures of antibodies made in a lab rather than taken from a recovered person’s blood.
“This approach predates a lot of the current science on vaccines in immunology and the antibody response,” Akselrod said. “For more than 100 years, people have tried this approach — us[ing] the blood of people recovered from an infectious disease to cure those still suffering from it. The promise is that there is historic experience with it. The downside is, of course, we don’t know how effective it will be for this virus, in part because there might be slight variations in the virus that circulates across the big population. Viruses do mutate and change their surface appearance in ways that try to evade the immune system.”
Clinical trials: Studies have been mixed. In October, the NIH wrote that there was insufficient evidence to recommend the use of convalescent plasma. However, a study published on January 6, 2021, in the New England Journal of Medicine showed that the treatment can reduce the risk of severe illness when given to patients over 65 within 72 hours of COVID-19 symptoms starting.
Most studies so far have not shown a benefit of using convalescent plasma. Additionally, monoclonal antibodies are being used regularly instead. In March 2021, the FDA halted a trial because, while the treatment appears to be safe, it showed no benefit to patients with mild symptoms.
The post Off-Label: Can Drugs Developed For Other Diseases Treat COVID-19? appeared first on MedShadow.
Original post here:
Off-Label: Can Drugs Developed For Other Diseases Treat COVID-19?